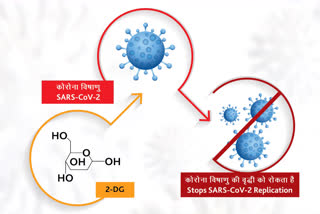
New Delhi:The Drugs Controller General of India (DCGI) has approved the emergency use of an anti-COVID drug - 2-deoxy-D-glucose (2-DG) - as an adjunct therapy in moderate to severe COVID-19 cases.
According to Defence Research and Development Organisation (DRDO) statement, an anti-COVID-19 therapeutic application of the drug 2-deoxy-D-glucose (2-DG) has been developed by the Institute of Nuclear Medicine and Allied Sciences (INMAS), a lab of DRDO in collaboration with Dr Reddy's Laboratories, Hyderabad.
Clinical trial results have shown that this molecule helps in faster recovery of hospitalized patients and reduces supplemental oxygen dependence. A higher proportion of patients treated with 2-DG showed RT-PCR negative conversion in COVID patients. The drug will be of immense benefit to the people suffering from Covid-19 in the ongoing pandemic, it added.
Earlier in April 2020, during the first wave of pandemic INMAS-DRDO scientists conducted laboratory experiments with the help of Centre for Cellular and Molecular Biology (CCMB), Hyderabad and found that this molecule works effectively against SARS-CoV-2 virus and inhibits viral growth.
Read:Mamata demands universal COVID vaccine programme; says never supports violence
Based on these results, DCGI- CDSCO permitted Phase-II clinical trial of 2-DG in COVID-19 patients in May 2020.